Case Study: Spatially Resolving Lithiation in Si-graphite Composite Electrodes via in situ High-resolution XRD-CT
Companies involved:
Finden Ltd, National Renewable Energy Laboratory, University College London, The Faraday Institution, Research Complex at Harwell, ESRF
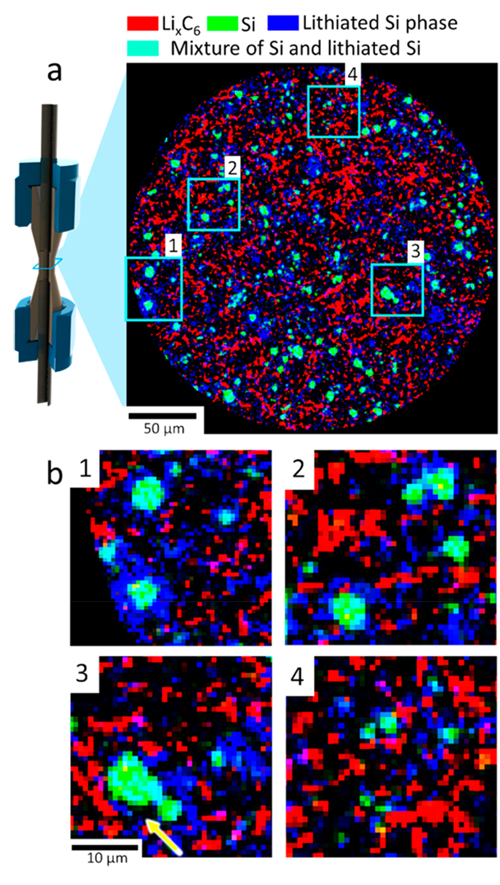
Figure 5. XRD-CT view of the Si−graphite electrode. (a) XRD-CT slice taken at the beginning of the charge step showing a phase-distribution map of LiC12 (red), crystalline Si (green), and lithium silicides LixSi (blue). According to additive color mixing, the color teal represents a mixture of green (Si) and blue (lithiated Si). (b) Magnified regions of interest showing large particles of LixSi phase with crystalline Si cores (1−3) and smaller LixSi particles (4) interspersed in the graphite matrix. The yellow arrow highlights what looks to be evidence of delamination from the crystalline Si core.
Challenge:
Optimizing chemical and morphological parameters of lithium-ion (Li-ion) electrodes is extremely challenging; in part due to the absence of techniques to construct spatial and temporal descriptions of chemical and morphological heterogeneities. The majority of mechanisms that lead to degradation and performance loss of Li-ion cell electrodes can be characterized by chemical, crystallographic and morphological heterogeneities that are present or develop during operation[1]. Particle and electrode cracking can arise from heterogeneous strains present in the active material or electrode matrix. The morphological evolution of electrode particles during operation can lead to the development of local strains, causing particle cracking and electrode delamination. Non-uniform particle sizes and spatial distributions of conductive carbon within an electrode can lead to some regions of the electrode being underutilized or degrading at an accelerated rate relative to the bulk. Understanding the link between cell performance and dynamic chemical, crystallographic, and morphological heterogeneities within electrodes is paramount to engineering cells that are optimised for specific operating conditions.
Sample:
Li ion battery cell with Si-Graphite electrode.
Solution:
We contributed to the first demonstration of combined high-speed X-ray diffraction (XRD) at 100 Hz and XRD computed tomography (XRD-CT) to probe, in 3D, crystallographic heterogeneities within Li-ion electrodes with a spatial resolution of 1 μm. The local charge transfer mechanism within, and between individual particles was investigated in a graphite-silicon (Si) composite electrode. High-speed XRD revealed charge balancing kinetics between the graphite and Si during the minutes following the transition from operation to open-circuit. Sub-particle lithiation heterogeneities in both Si and graphite were observed using high-resolution XRD-CT.
Benefits:
X-ray diffraction (XRD) has been extensively used to characterize the phases of intercalation materials, such as graphite, ex- or in situ[9, 10]. Early in situ experiments[9] involved scans that were carried out while the cell was at open circuit, had long exposure times, and relied on bulk electrode measurements. With the advancement of high-flux synchrotron sources, focusing optics and detectors, high-resolution point scans are now feasible in short amounts of time, facilitating spatially-resolved operando diffraction measurements. Synchrotron XRD has been used to monitor the activity of distinct electrode chemistries in layered composite positive electrodes and to construct depth profiles of lithiation within a Si electrode, by recording point scans from separator to current collector and monitoring the phase change from crystalline Si to amorphous phase during its first lithiation [2] [3].
Using XRD-CT to identify 3D crystallographic heterogeneities in the bulk electrodes has recently been demonstrated in Ni-metal hydride and sodium-ion cells[4-6]. High resolution (> 1 μm) XRD-CT scans have been achieved but acquisition times have been on the order of many hours[7, 8]. In this work, we leveraged the unparalleled time-resolved, high-resolution XRD-CT capabilities at Beamline ID15A at The European Synchrotron[9, 10] to investigate dynamic processes in 3D with micron-scale resolution. For the first time, spatial heterogeneities within single electrode particles (Si and graphite) were identified. This demonstrates a significant step forward in spatially and temporally resolving states of charge and degradation phenomena within electrode particles during operation and is expected to be a platform study for further investigations into chemical heterogeneities in operating electrochemical cells.
Further reading:
Spatially Resolving Lithiation in Silicon–Graphite Composite Electrodes via in Situ High-Energy X-ray Diffraction Computed Tomography, Donal P. Finegan, Antonis Vamvakeros, Lei Cao, Chun Tan, Thomas M.M. Heenan, Sohrab R. Daemi, Simon Jacques, Andrew M. Beale, Marco Di Michiel, Kandler Smith, Paul R. Shearing, Chunmei Ban, May 2019, Nano Letters.
References:
[1] S.J. Harris, P. Lu, The Journal of Physical Chemistry C, 117 (2013) 6481-6492.
[2] P. Pietsch, M. Hess, W. Ludwig, J. Eller, V. Wood, Scientific Reports, 6 (2016) 27994.
[3] T. Sasaki, C. Villevieille, Y. Takeuchi, P. Novák, Advanced Science, 2 (2015) 1500083.
[4] G. Harding, J. Kosanetzky, U. Neitzel, Medical Physics, 14 (1987) 515-525.
[5] J. Sottmann, M. Di Michiel, H. Fjellvåg, L. Malavasi, S. Margadonna, P. Vajeeston, G.B.M. Vaughan, D.S. Wragg, Angewandte Chemie International Edition, 56 (2017) 11385-11389.
[6] K.M.Ø. Jensen, X. Yang, J.V. Laveda, W.G. Zeier, K.A. See, M.D. Michiel, B.C. Melot, S.A. Corr, S.J.L. Billinge, Journal of The Electrochemical Society, 162 (2015) A1310-A1314.
[7] P. Bleuet, E. Welcomme, E. Dooryhée, J. Susini, J.-L. Hodeau, P. Walter, Nature Materials, 7 (2008)
468.
[8] A. Bonnin, J.P. Wright, R. Tucoulou, H. Palancher, Applied Physics Letters, 105 (2014) 084103.
[9] S.D.M. Jacques, M. Di Michiel, A.M. Beale, T. Sochi, M.G. O’Brien, L. Espinosa-Alonso, B.M. Weckhuysen, P. Barnes, Angewandte Chemie International Edition, 50 (2011) 10148-10152.
[10] A. Vamvakeros, S.D.M. Jacques, M. Di Michiel, D. Matras, V. Middelkoop, I.Z. Ismagilov, E.V. Matus, V.V. Kuznetsov, J. Drnec, P. Senecal, A.M. Beale, Nature Communications, 9 (2018) 4751.
Read more about our team and further publications.
